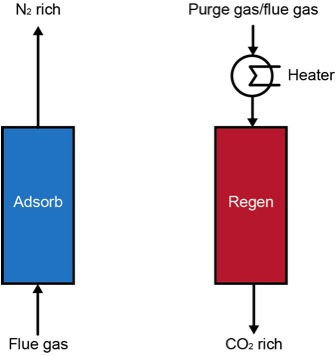
__ The general design of an adsorption
carbon capture process involves a large, packed bed of solid adsorbents. The product flue gas is passed through the
bed, where the CO2 is adsorbed by the confined MOF, exiting the bed
with significantly less CO2.
This exit stream is recycled through the bed in maximize the CO2
uptake. The CO2 loaded bed is
then heated to a critical temperature at which the captured CO2 is
desorbed/released as a high purity stream of CO2, which is
subsequently compressed for transportation and storage. The following figure summarizes and details
the continuous two bed process.
Figure 1. Two bed process design for a temperature-swing adsorption (TSA) setup
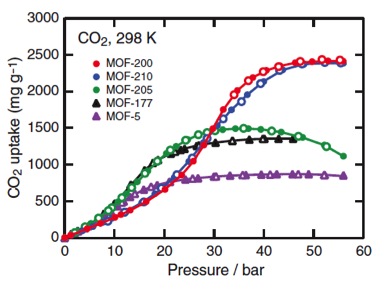
It follows that the total cost for a proposed plant can be separated into capital and operational costs. The capital cost is variable, a direct function of the capacity of the selected adsorbent because of its implications with regard to the adsorbent price, necessary amount and bed vessel for a desired removal of CO2. Likewise, the operational cost is largely dependent on variable factors that dictate the required amount of parasitic energy necessary for the process. The excitement revolving around MOF adsorption can be largely attributed to two of their defining, unique characteristics that have major implications to their economic outlook and future feasibility: a high surface area and chemical tunability. Maximizing the uptake of CO2 in MOFs is achieved by increasing the number of adsorptive sites within the given material. This is easily attained by an increase in material surface area, resulting in an increased material porosity. Generally speaking, increasing the surface area of a MOF is as easy as expanding the organic molecular links. This is simpler in theory than application because expanding the organic links comes at the expense of the structural integrity of the framework. However, some MOFs have been synthesized with specific regard to an increased surface area and have yielded impressive implications. By successfully synthesizing MOFs with heightened surface areas, the CO2 adsorption capacity has been expanded as the following figure supports: _Figure 2. CO2
adsorption isotherms, including that for MOF-210. Note that MOF-210 only
attains its full capacity at high CO2 pressures (1).
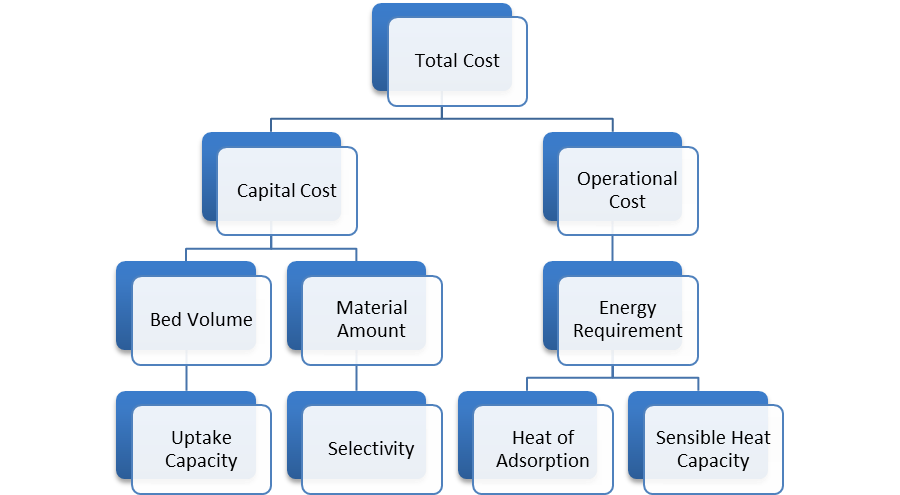
Although CO2 uptake remains relatively low at lower pressures, which are characteristic of post-combustion flue gas, it still represents a noted improvement. An increased uptake allows for a decrease in both capital and operational costs. This increase will require less material for a given CO2 removal, which also allows for a decreased bed size. Both implications will see a noted decrease in the design capital cost. The overall cost is even further decreased because of MOFs chemical tunability. In carbon capture via adsorption, as is the case with other methods of separation like absorption or membranes, the overall total cost of the process is a combination of capital and operational costs. Each method of separation revolves around a different process for separation. As such, different characteristic process variables exist for each method, which causes variation in the effective total process cost. For adsorption, the resulting total cost is dependent on the following process variables: Figure 3. Total cost flow chart
_
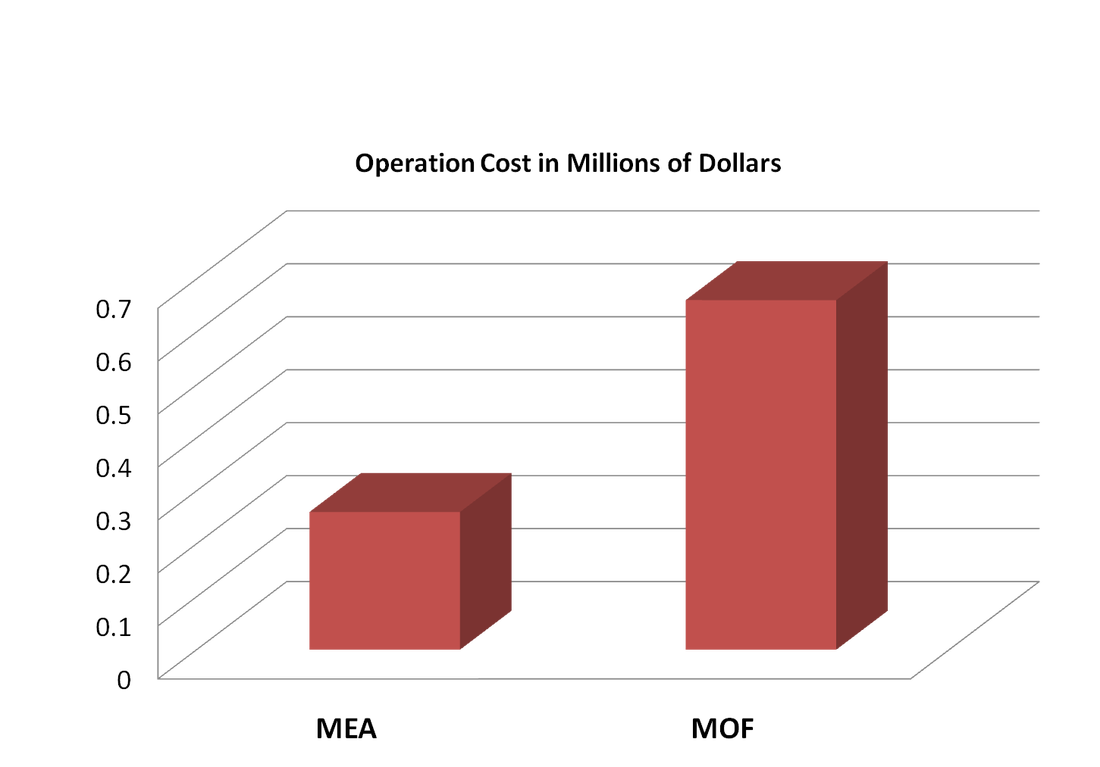
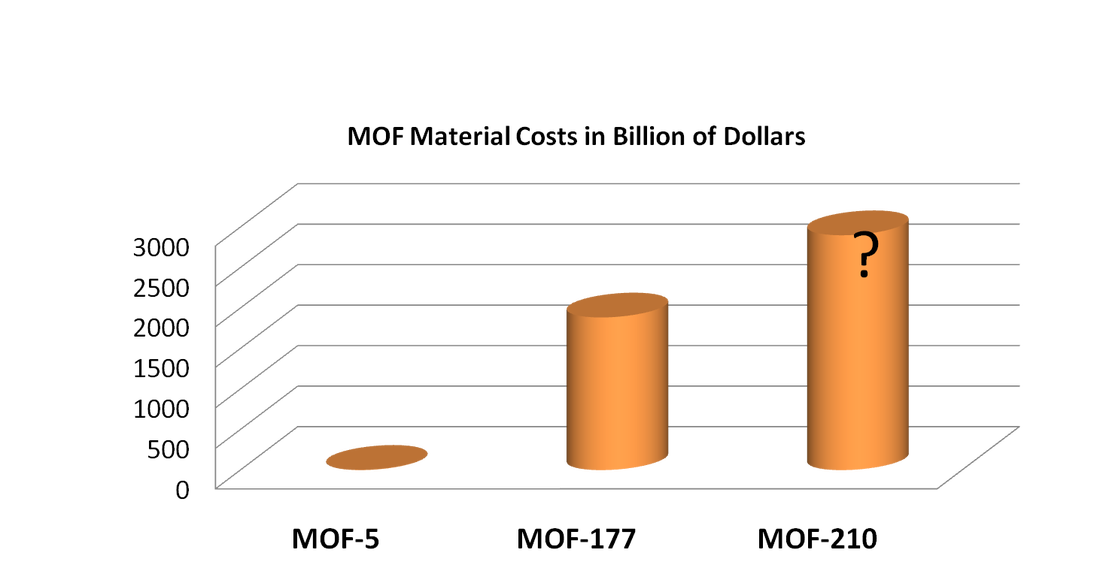
Unfunctionalized solid surfaces typically adsorb CO2 via a physisorption mechanism. However, the introduction of functional groups in a MOF can increase the selective adsorption of CO2, as is the case with the recently published amine-functionalized MOF research. Higher selectivity existing in the material translates to increased adsorption capacity, thereby reducing the amount of necessary material and associated capital costs. However, the chemical tunability of MOFs, the ability to modify the material to optimize favorable conditions, can also impact the operational costs, which are largely a product of the required parasitic energy. Because the adsorption separation cycle operates between the different temperatures of adsorption and desorption, a large fraction of the parasitic energy concerns the cyclic heating and cooling of the material. Thus, tuning a MOF that has an optimal heat of adsorption can minimize the parasitic energy and operational cost. For the greatest effect, a MOF should be tuned to maximize the adsorption capacity for CO2 in the flue gas, which should be tailored to the low pressure (~0.15 bar) and temperature specified by the flue gas. Additionally, a desired MOF should exhibit a low heat of adsorption to sizably reduce the energy that is needed to heat and cool the material. Cost for MOF TSA design is mainly decided by its parasitic energy, though we will see that the initial one-off investment is just too high comparing to other costs. Giving assumed condition of our design, two columns of MOF working at 14hr cycle will require power input of 220MW from the plant. Considering its only a 500MW coal-fire power plant, that's quite some input. With current electricity price for industry from PG&E, we are paying $0.66M per day to capture and store CO₂ emitted (2). Comparing to MEA absorption technique (see Figure 4), which is more mature than MOF adsorption and cost only $0.26M per day from parasitic energy, we can say MOF is appealing, but not appealing enough unless we can reduce the energy we have to divert to it. For our TSA design, equipments like columns, heat exchangers, etc, will cost within a million, pretty standard for industry. Maintainance of MOF adsorber is mainly taking care of the equipment, since the solid adsorbent doesn't neeeds to be replaced, unlike MEA where we need to constantly replenish absorbent. So it seems that MOF design has a lower capital cost in the long run, and would be more attracting if we consider the environmental impact from MEA. But, the initial setting up is so expensive that it is not feasible at all. Giving the price of MOF-5, where we assumed MOF-210 will eventually cost, filling two columns will cost us $2.9B. Just for reference, MOF-5 costs $200/kg, MOF-177 costs $130K/kg, and MOF-210 is assumed to be even higher (see Figure 5), comparing to MEA which only costs $2.66/kg (3,4). Just to imagine, a 500MW coal-fire power plant needs to spend $2.9B to capture its CO₂ and reduces its power output to 300MW, we can see this TSA design is not feasible at all. In the future, we can improve our selectivity and adsorbing capability for MOF TSA design to be more feasible, also we need to reduce the price of synthesising MOF at industry scale to mass deploy them. Giving current track of alternative energy power station, MOF CCS has a long way to go. Figure 4. Comparison of parasitic energy cost for typical MEA absorption and MOF adsorption for a 500 MW coal-fired power plant
Figure 5. Comparison of material costs for different types of MOFs. Note that the cost of MOF-210 is unknown.
|